Packing Size:
20 pcs/box 、50 pcs/box
100 pcs/box

SpecimenType:
Nasopharyngeal secretion
Throat secretion
Anterior nasal secretion

Detection Time:
15-20minutes

Shelf Life:
24 months

Storage:
2-30C°

Packing Size:
20 pcs/box
50 pcs/box 、100 pcs/box

Storage:
2-30C°

Detection Time:
15-20minutes

Specimen type:
Nasopharyngeal secretion
Throat secretion
Anterior nasal secretion

Shelf Life:
24 months
Watch us
Watch us
MEDOMICS
MEDOMICS

SARS-CoV-2
Antigen Test Kit(LFIA)
Nasopharyngeal Swab
Throat Swab
Anterior Nasal Swab
Determine the infection of someone by
detecting viral material.
SARS-CoV-2 Antigen Test Kit(LFIA)
Nasopharyngeal Swab Throat Swab Nasal Swab
Determine the infection of someone bydetecting viral material.
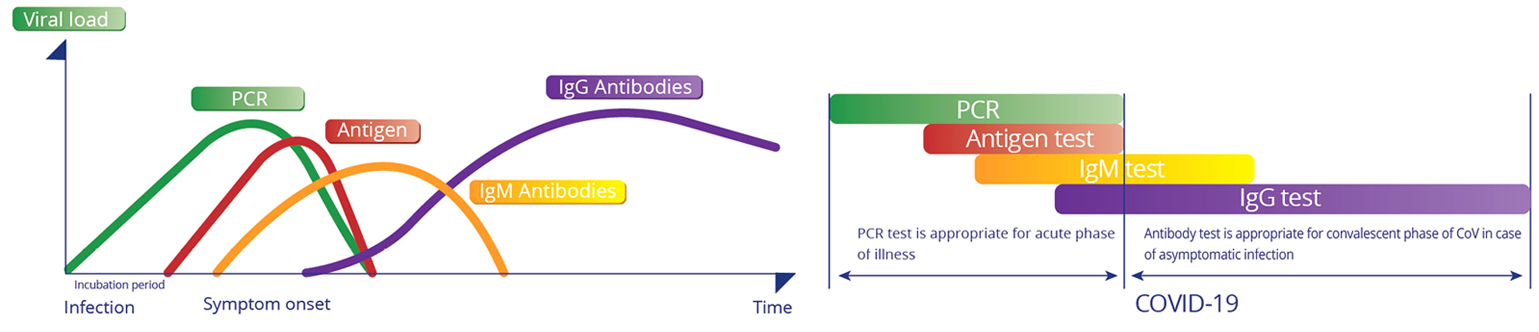
Test design and principle
Test design and principle
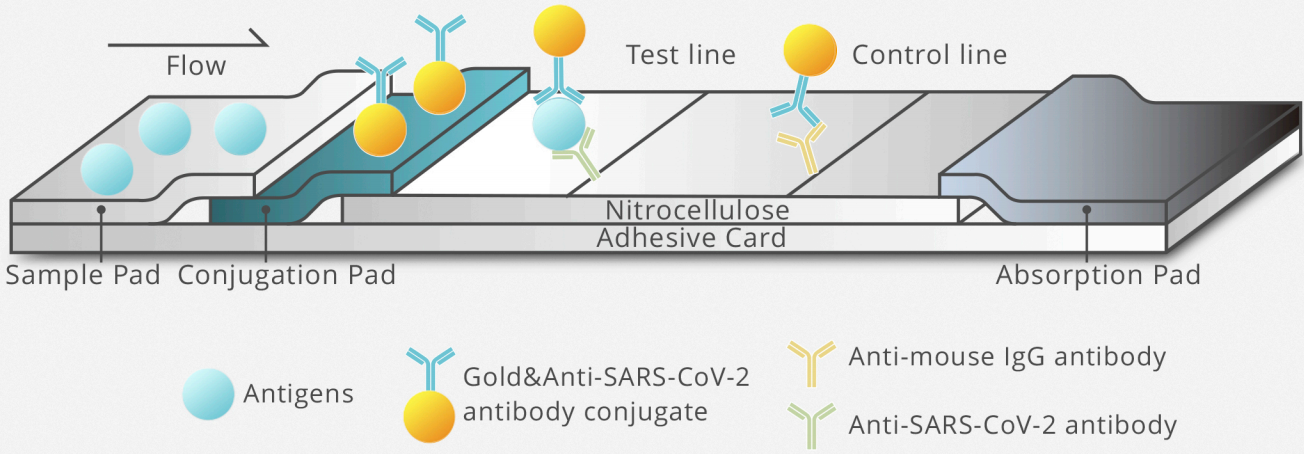
Colloidal gold-labeled anti-SARS-CoV-2 antibody is sprayed on conjugation pad.
Test line is fixed with anti-SARS-CoV-2 antibody for detecting the novel coronavirus.The quality control antibody is fixed on the control line.
Colloidal gold-labeled anti-SARS-CoV-2 Antibody
is sprayed on conjugation pad.
Test line is fixed with anti-SARS-CoV-2 Antibody
for detecting the novel coronavirus.The quality control antibody is fixed on the Control line
Sensitivity and Specificity
The Medomcis SARS-CoV-2 Antigen Test Kit (LFlA) was evaluated with different clinical sam-ples whose status were confirmed by RT-PCR.The results are shown in following tables.
Sensitivity and Specificity
The Medomcis SARS-CoV-2 Antigen
Test Kit (LFlA) was evaluated with different clinical
sam-ples whose status were confirmed by RT-PCR.
The results are shown in following tables.
1.Table 1 Nasopharyngeal or throat secretion collection

2.Table 2 Anterior nasal secretion collection
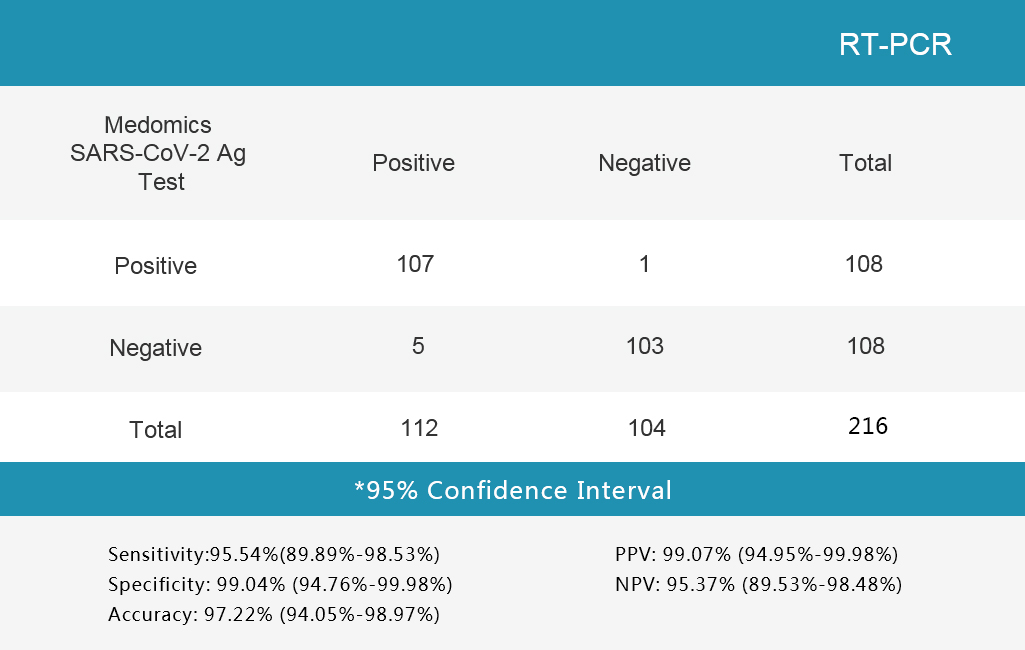
Sensitivity and Specificity
Sensitivity and Specificity

FOR IN VITRO
DIAGNOSTIC USE ONLY.
FOR PROFESSIONAL
USE ONLY.
FOR IN VITRO DIAGNOSTIC USE ONLY.

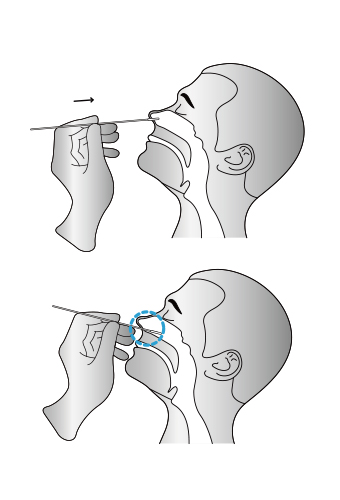
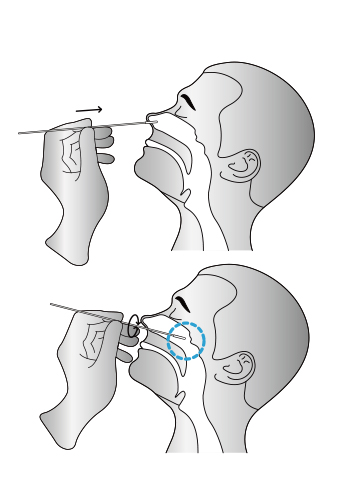
Sample Requirements
Sample Requirements
01
02
03
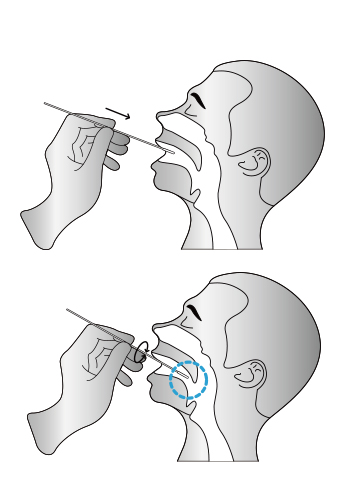
Insert the whole swab completely into the throat from the mouth, centering on the throat wall and the
reddened area of the palate tonsil, wipe both sides of the pharyngeal tonsil and posterior pharyngeal wall with moderate force. Try to avoid the tongue before taking it out.
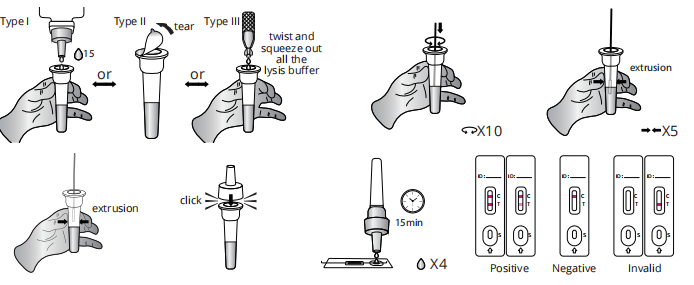
Test Procedure
Test Procedure
Do not open pouch until ready to use. Prep necessary materials: Timer | Tube rack for sampling tubes and specimens | Any necessary personal protective equipment.
1 | Sampling: vertically add 15 drops (approximately 350 μL) lysis buffer into the sampling tube from vial or open the seal of the sampling tube containing lysis buffer or twist and squeeze out all the lysis buffer into the sampling tube from capsule. Insert the swab (aftercollection) into the buffer. Rotate the swab against the inner tube wall 10 times and squeeze the swab from the outer tube wall 5 times to completely dissolve the sample in the buffer, the outer tube wall in order to leave the sample in the tube as much as possible.Remove and discard the swab, cover the tube with the dropper.
2 | Test procedures: Open the aluminum foil pouch, take out the test cassette and lay it on a clean flflat surface, then mark the cassette with the patient ID or sample number and add 4 drops (approximately 100 μL) processed sample extract into the sample well.The result should be observed within 15-20 minutes. Results observed after 20 minutes are invalid.
DISPLAY OF THE RESULT / EXPECTED VALUE
DISPLAY OF THE RESULT / EXPECTED VALUE
“C”:Quality Control Line
“T”:Detection Line
“S”:Sample Well
“C”:Quality Control Line
“T”:Detection Line
“S”:Sample Well
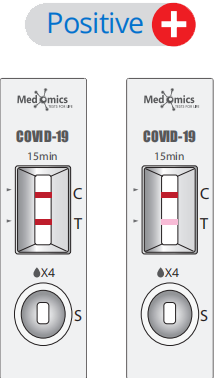
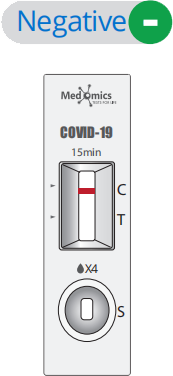
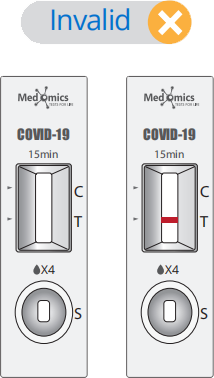
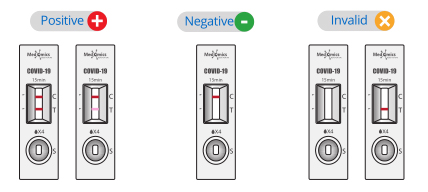
Display of Results/Expected Values
Display of Results/Expected Values
• Negative result: If only the quality control C line appears and the detection T line is not visible, the sample contains no SARS-CoV-2 antigens or the SARS-CoV-2 antigens concen tration is lower than the limit of detection and the result is negative.
• Positive result: If both the quality control C line and the detection T line appear, then theSARS-CoV-2 antigens have been detected and the result is positive.
• Invalid result: If the C line does not appear, the result is invalid and a new test must be performed.
Note: The color intensity of the T line is related to the concentration of SARS-CoV-2 Antigens contained in the sample, and the result should be determined by whether the Tline is colored or not regardless of the color intensity.
External Positive and Negative Controls
External Positive and Negative Controls
The external controls consist of positive swab and negative swab are available. Only one red colored C line in the observation window can be seen while using a negative control swab. Both red colored C line and T line in the observation window can be seen while using a positive control swab. If necessary, please contact your local vendor or Medomics to obtain control swabs.
Related Products
Read more +
Related Products
Read more +
Search for more you need
Enter keywords, you can query more product information you would like to know.
Contact Information
E-mail:
Address:Building 01, Phase 6, No.71, Xinghui Road, Jiangbei New Area, Nanjing
About Us
Product Center
Jiangsu Medomics Medical Technology Co,Ltd Powered By www.300.cn 苏ICP备18065762号